|
 |
- Search
| J Electrodiagn Neuromuscul Dis > Volume 26(1); 2024 > Article |
|
Abstract
Colchicine is a widely used anti-inflammatory medication, but its neuromuscular adverse effects are under-recognized. One month ago, a 70-year-old woman presented to our clinic for an evaluation of general weakness. She had been taking colchicine and diuretics daily. The weakness, which began in both thighs and lower legs approximately 4 weeks before her visit, had progressively worsened. The patient also experienced mild paresthesia and hypoesthesia in both arms and legs. Her serum creatine kinase and aldolase levels were elevated, and nerve conduction studies indicated a motor-dominant sensorimotor polyneuropathy of the axonal type. Needle electromyography showed prominent fibrillation potentials, positive sharp waves, and myotonic discharges. Suspecting colchicine-induced neuromyopathy, we discontinued the colchicine and diuretics, after which her symptoms resolved.
Colchicine is an anti-inflammatory medication used to treat various inflammatory conditions, such as familial Mediterranean fever and acute gouty arthritis [1]. Common side effects of colchicine include gastroenteritis, blood dyscrasias, and dermatitis. However, the neuromuscular adverse effects associated with colchicine are not well recognized [2]. Patients on concurrent medications or those with underlying renal insufficiency may face an elevated risk of experiencing neuromuscular complications from colchicine, which include chronic neuromyopathy and acute fulminant rhabdomyolysis. The characteristic neuromuscular complication induced by colchicine is neuromyopathy, which manifests as an elevated serum creatine kinase (CK) level, subacute weakness in the proximal lower extremities, and length-dependent sensory peripheral neuropathy, particularly when combined with renal impairment [3]. We report a case of colchicine-induced neuromyopathy with myotonic discharges in a patient using concomitant diuretics.
One month ago, a 70-year-old woman presented to our clinic for an evaluation of general weakness. Her extensive medical history included hypertension, heart failure with pericardial effusion, hypothyroidism, and hyperuricemia. The weakness began in her thighs and lower legs approximately 4 weeks before her visit and had progressively worsened. She displayed mild myalgia along with slight paresthesia and hypoesthesia in both arms and legs. The patient did not indicate any cranial nerve deficits or issues with bladder or bowel function. However, she did report persistent diarrhea and poor oral intake over the prior 3 weeks. She had been taking colchicine at a dose of 1.2 mg daily for several months to manage hyperuricemia and pericardial effusion. Additionally, she had been on diuretics, including furosemide at 80 mg and spironolactone at 25 mg, for 2 months to address generalized edema and pleural effusion. Her medication regimen also included levothyroxine at 0.15 mg, aspirin at 100 mg, and allopurinol at 100 mg.
The initial physical examination revealed an arterial blood pressure of 133/69 mm Hg, a pulse rate of 98 beats per minute, a respiratory rate of 19 breaths per minute, and a body temperature of 36.6°C.
Laboratory test results indicated anemia, with a hemoglobin level of 8.2 g/dL (normal range, 13.5 to 17.5), and thrombocytopenia, with a platelet count of 55,000/µL (normal range, 150,000 to 400,000). Elevated liver enzymes were also noted, including alkaline phosphatase at 104 U/L (normal range, 7 to 45) and aspartate aminotransferase at 93 U/L (normal range, 8 to 43). Additionally, the patient’s blood urea nitrogen level was elevated (33 mg/dL; normal range, 10 to 20), as was her creatinine level (1.8 mg/dL; normal range, 0.6 to 1.1). Her serum CK level was high, at 2,581 U/mL (normal range, 52 to 336), and her aldolase level was also high, at 9.4 U/mL (normal range, 1 to 7.5).
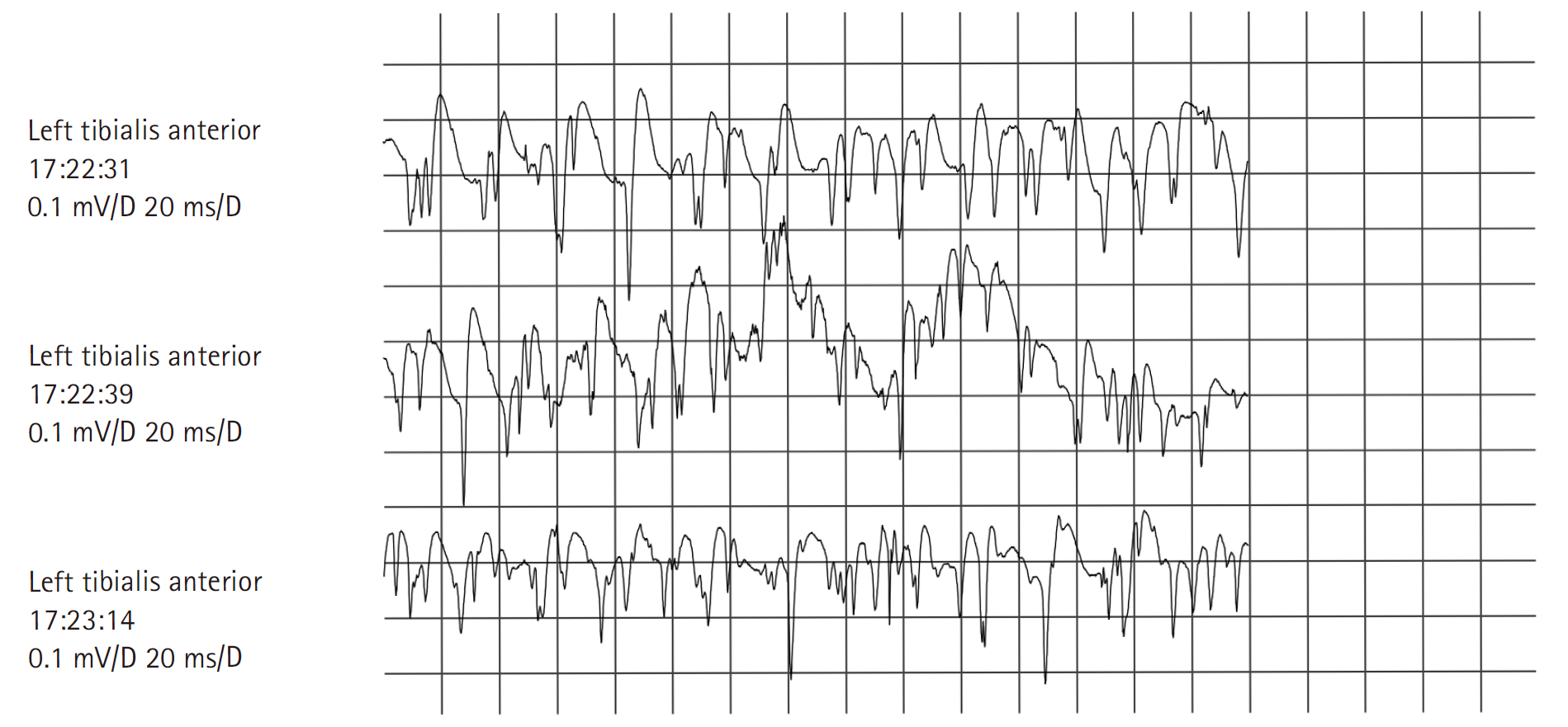
On neurological examination, the patient’s cognitive functions and cranial nerves appeared normal. She exhibited both proximal and distal upper limb weakness (Medical Research Council [MRC] grade 3/5), severe weakness in hip flexion and ankle dorsiflexion (MRC grade 2/5), and diffuse weakness in the lower limbs (MRC grade 3/5). No clinical myotonia, muscle atrophy, or fasciculations were observed. Deep tendon reflexes were diffusely reduced in the upper limbs and absent in the legs. The patient was experiencing graded sensory loss, with pain and a cold sensation present up to the knees, reduced vibratory sensation at the ankles, and diminished joint position sensation in the toes. Our initial clinical diagnosis was Guillain-Barré syndrome. Motor nerve conduction studies revealed prolonged terminal latencies, low-amplitude compound muscle action potentials, and slow motor nerve conduction velocities in both median, ulnar, posterior tibial, and peroneal nerves, with asymmetrical involvement (Table 1). Sensory nerve conduction studies indicated low-amplitude action potentials and slightly slow conduction velocities in both median nerves (Table 2). These studies suggested a motor-dominant sensorimotor polyneuropathy of the axonal type. However, needle electromyography (EMG) demonstrated prominent fibrillation potentials, positive sharp waves, and myotonic discharges in the proximal and distal limb muscles (Table 3, Fig. 1). Analysis of motor unit action potentials revealed short duration and early recruitment in proximal muscles, but normal duration and reduced recruitment in distal muscles (Table 3). Cerebrospinal fluid studies and anti-ganglioside antibody tests for Guillain-Barré syndrome fell within normal ranges. Tests for anti-nuclear antibody, anti-double-stranded DNA (deoxyribonucleic acid) antibody, anti-Jo-1 antibody, anti-SCL-70 antibody, and anti-centromere antibody, which are used in the diagnosis of inflammatory myositis, were also within normal limits. Thyroid function tests and anti-thyroid peroxidase antibody titers were normal. Cytosine-thymine-guanine (CTG) repeat analysis of the dystrophia myotonica protein kinase (DMPK) gene, used in the diagnosis of myotonic dystrophy, was within the normal range as well. We suspected toxic or drug-induced neuromyopathy, as the electrophysiological findings could be attributed to the simultaneous occurrence of acute polyneuropathy and myopathy. The clinical, laboratory, and electrophysiological findings were consistent with colchicine-induced neuromyopathy exacerbated by diuretics in the context of renal insufficiency. Upon admission, colchicine and diuretics were discontinued, and within 1 week, the patient’s diarrhea, anemia with thrombocytopenia, and renal insufficiency resolved. After 2 months, she exhibited no weakness in the arms and only mild weakness in the lower limbs (MRC grade 4/5). Six months later, her nerve conduction study and serum CK levels were normal (Tables 4, 5).
Written informed consent by the patients was waived due to a retrospective nature of our study.
By binding to tubulin, a crucial protein in mitosis, colchicine inhibits microtubule polymerization and prevents microtubule elongation [1]. Complications of colchicine treatment typically begin with gastrointestinal symptoms, such as abdominal pain, nausea, vomiting, and diarrhea, within 24 hours after the oral ingestion of large amounts [4]. In our patient, chronic diarrhea preceded the onset of neuromuscular symptoms. These gastrointestinal side effects should be managed to prevent dehydration, hypovolemic shock, and cardiovascular collapse. However, the present patient’s continued use of diuretics exacerbated dehydration and led to renal dysfunction. The mechanisms underlying colchicine-associated neuromuscular complications are not fully understood and are often overlooked. Nonetheless, colchicine-induced neuromyopathy is frequently observed in patients taking the standard dose, and certain conditions significantly increase the risk. These include impaired renal and hepatic function, as well as the concurrent use of medications that inhibit the cytochrome P450 3A4 isozyme (CYP3A4) and P-glycoprotein [5]. Patients who receive doses within the usual range may be relatively susceptible to this neuromuscular complication if they have chronic renal dysfunction or take diuretics [2]. Colchicine-induced neuromyopathy can also result from the coadministration of any medication metabolized by the CYP3A4 system [6]. Previous reports have indicated that numerous drugs, including clarithromycin, pravastatin, simvastatin, fluvastatin, atorvastatin, gemfibrozil, calcium channel blockers, and cyclosporine, can contribute to neuromyopathy when they interact with colchicine [7].
Our patient exhibited no neuromuscular issues while on colchicine monotherapy. However, the addition of diuretics led to renal insufficiency, which precipitated colchicine-induced neuromyopathy. Often, the primary clinical feature of this condition is painless proximal lower limb weakness; nevertheless, myalgia can sometimes be observed, as with our patient [8]. Additional manifestations, such as distal leg weakness, arm weakness, sensory abnormalities, and areflexia, have also been reported and were present in this case [8]. The course of colchicine-induced neuromyopathy is generally subacute, spanning 1 to 3 months, but the condition can present acutely in less than 4 weeks or chronically over more than 3 months [2]. While no respiratory or cardiac complications have been documented, dysphagia has been noted as an occasional symptom [2]. Laboratory tests typically reveal mild to marked elevation of CK levels [2]. Electrodiagnostic studies may suggest a sensorimotor axonal polyneuropathy and myopathic motor unit potentials with signs of membrane irritability, similar to findings observed with inflammatory myopathies [3]. Due to the predominant proximal weakness, elevated CK level, and irritable myopathy on EMG, polymyositis is frequently considered in the differential diagnosis for patients with colchicine-induced neuromyopathy. Although rare, the literature includes instances of colchicine-induced myopathy or neuromyopathy with myotonic discharges on EMG, yet without clinical myotonia [9]. In rare cases, colchicine alone has been implicated in peripheral neuropathy with electrical myotonia [10]. We contend that the presence of a myopathic pattern with myotonic discharges on EMG in patients taking colchicine is a noteworthy finding that may lead to unnecessary invasive muscle biopsy. Histologically, muscle tissue frequently exhibits vacuolar myopathy with increased acid phosphatase reactivity, which is particularly pronounced in proximal muscles [2]. The disruption of the microtubule-dependent cytoskeletal architecture is considered a potential pathogenic mechanism for colchicine-induced neuromyopathy [2]. A thorough patient history can help exclude neuromyopathy resulting from exposure to drugs or toxins such as alcohol, amiodarone, chloroquine, and vincristine.
Most patients who discontinue colchicine experience a complete reversal of symptoms [5]. Withdrawal from colchicine typically leads to a rapid improvement in the clinical and electrophysiological abnormalities associated with myopathy, although the resolution of neuropathy can take longer [3]. Colchicine-induced neuromyopathy should be considered in patients—particularly elderly individuals with renal insufficiency or those taking the drug in combination with other medications—who present with subacute weakness in the proximal lower extremities and distal sensory changes, even if electrophysiological studies indicate polyneuroradiculopathy and/or active myopathy with or without myotonic discharges. Early recognition of the characteristic features of colchicine-induced neuromyopathy can help avoid unnecessary muscle biopsies. Most cases involving clinical weakness and laboratory abnormalities can be resolved promptly with cessation of the medication.
Table 1.
Nerve Conduction Study (Initial): Motor Nerve Conduction Study
Table 2.
Nerve Conduction Study (Initial): Sensory Nerve Conduction Study
Table 3.
Needle Electromyographic Findings (Initial)
Table 4.
Nerve Conduction Study (6 Months After Discontinuation): Motor nerve conduction study
Table 5.
Nerve Conduction Study (6 Months After Discontinuation): Sensory Nerve Conduction Study
References
1. Altman A, Szyper-Kravitz M, Shoenfeld Y: Colchicine-induced rhabdomyolysis. Clin Rheumatol 2007;26:2197-2199.


2. Kuncl RW, Duncan G, Watson D, Alderson K, Rogawski MA, Peper M: Colchicine myopathy and neuropathy. N Engl J Med 1987;316:1562-1568.


3. Kuncl RW, Cornblath DR, Avila O, Duncan G: Electrodiagnosis of human colchicine myoneuropathy. Muscle Nerve 1989;12:360-364.


5. McEwan T, Bhambra J, Liew DF, Robinson PC: Systematic review of colchicine neuromyopathy: risk factors, duration and resolution. Semin Arthritis Rheum 2023;58:152150.


6. Mor A, Mitnick HJ, Pillinger MH, Wortmann RL: Drug-induced myopathies. Bull NYU Hosp Jt Dis 2009;67:358-369.

7. Kwon YH, Kang HJ, Lee JH, Kim DH, Hwang M: A case of colchicine induced acute myopathy in atorvastatin and calcium channel blocker user. J Korean EMG Electrodiagn Med 2009;11:169-173.

-
METRICS

-
- 0 Crossref
- Scopus
- 202 View
- 1 Download
- Related articles in J Korean Assoc EMG Electrodiagn Med
-
A Case of Acute Colchicine-induced Polyneuropathy with Myotonia in a Gout Patient2013 December;15(2)