 |
 |
- Search
| J Electrodiagn Neuromuscul Dis > Volume 24(3); 2022 > Article |
|
Abstract
Thoracic outlet syndrome (TOS) is caused by the compression of neurovascular structures in the thoracic cage. TOS can be classified into neurogenic TOS (NTOS) and vascular TOS. Congenital anomalies, such as cervical ribs and elongated C7 transverse processes, may be the cause of NTOS. NTOS can be subcategorized as either disputed NTOS or true NTOS. True NTOS, a very rare disease with a prevalence of about one in a million, is diagnosed by the weakness and atrophy of T1-innervated intrinsic muscles of the hand and corresponding electrodiagnostic abnormalities. We report a case of a 42-year-old patient, who presented with a 5-year history of clumsiness and 1-year history of weakness and atrophy in her right hand. The patient was diagnosed with true NTOS during comprehensive rehabilitation, which she received for left hemiplegia due to intracranial hemorrhage. Elongated bilateral C7 transverse processes were found on a radiological evaluation. True NTOS is often overlooked or mistaken for carpal tunnel syndrome, thus delaying the diagnosis and inevitably worsening outcomes. Therefore, an electrodiagnostic study is crucial for detecting true NTOS, rather than typical physical examinations such as the Roos stress test and Adson’s test.
Thoracic outlet syndrome (TOS) is a complex of symptoms caused by the compression or irritation of neurovascular structures in the thoracic cage [1]. TOS can be classified into neurogenic TOS (NTOS) and vascular TOS. NTOS is a compressive peripheral neuropathy—specifically, a type of entrapment neuropathy caused by the compression of the brachial plexus. NTOS accounts for 95% of all TOS [1]. Congenital anomalies found in the cervical ribs and elongated C7 transverse processes may be the cause of NTOS [2]. NTOS can be subcategorized as either disputed NTOS or true NTOS [3]. Disputed NTOS occurs in a large group of patients with unexplained pain in the brachial, scapular, and cervical regions, with a rate that has been widely quoted as 3 to 80 per 1,000 people [3,4]. Disputed NTOS requires a clinical diagnosis without standard diagnostic criteria that depends on symptoms and provocative maneuvers, such as the Roos stress test and Adson’s test, because nerve conduction studies and electromyography are commonly negative for disputed NTOS [1,3]. In contrast, true NTOS, a very rare disease with a prevalence of about one in a million, resulting from the compression of C8 and T1 roots (T1 dominant) or proximal lower trunk of the brachial plexus, is diagnosed on the basis of objective weakness and atrophy of T1-innervated intrinsic muscles of the hand and corresponding electrodiagnostic abnormalities [5-7]. We report the case of a 42-year-old patient with progressive weakness and atrophic changes in the intrinsic muscles of the right hand, diagnosed with true NTOS during comprehensive rehabilitation after intracranial hemorrhage (ICH).
A 42-year-old woman was referred to our hospital with sudden-onset left-side weakness and an acute ICH of approximately 3.8 cm in the right basal ganglia. She was transferred to the Department of Rehabilitation Medicine after a month to start early comprehensive rehabilitation treatment. She demonstrated a full range of motion in all bilateral joints, with Medical Research Council (MRC) grade of 2 in her left upper and lower extremities. Meanwhile, atrophy and weakness of her right hand’s intrinsic muscles were remarkable (Fig. 1A, B), despite the lack of any lesion in the left brain on computed tomography (CT) scans. A physical examination revealed MRC grades of 0 in her right abductor pollicis brevis (APB) muscle and 2 in her abductor digiti minimi (ADM) and first dorsal interosseous (FDI) muscles. According to the patient, discomfort that disabled her from any fine movements in her right hand had gradually progressed for 5 years. She visited an orthopedic center at the first onset of symptoms and was informed of the possibility of carpal tunnel syndrome (CTS). An electromyographic (EMG) study revealed that a differential diagnosis for brachial plexopathy was necessary. However, based on her clinical symptoms, she was given an explanation of CTS and recommended to receive conservative treatment. The weakness and atrophy gradually progressed over the past years, making her daily activities, such as using a spoon, increasingly difficult, but were not aggravated within the last 3 months. No changes in the sensation of light touch and temperature were observed on her right side. She also did not complain of any pain in the upper arm or cervical regions. Special physical examinations for median nerve lesions including Phalen’s test, Tinel’s sign, and the hand of benediction were all negative. During the ulnar nerve examination, Wartenberg’s sign was positive in her right hand (Fig. 1C), whereas Froment’s sign and Tinel’s sign were negative. Additional examinations also revealed negative findings for the Roos stress test and Adson’s test. To determine the cause of the weakness and atrophy, electrodiagnostic studies were conducted using the Viking Select EMG NCS Machine (NICOLET EDX; Natus, Pleasanton, CA, USA). No compound motor action potential (CMAP) response in the right median nerve and a low amplitude of sensory nerve action potentials in the right ulnar and median antebrachial cutaneous (MABC) nerves were observed. The right median sensory nerve action potential was normal, and although the right ulnar CMAP amplitude was slightly decreased compared to that of the left side, it was still within the normal range (Table 1). In needle EMG, despite the lack of abnormal spontaneous activity, there was increased insertional activity, large motor unit action potentials (MUAPs), and reduced recruitment in the right C8 and T1-innervated muscles, including the extensor indicis proprius, abductor pollicis longus, flexor carpi ulnaris, FDI, and ADM. No MUAP was observed in the right APB muscle (Table 2). The EMG findings suggested right brachial plexopathy involving the lower trunk level, clinically consistent with NTOS. We consulted a thoracic surgeon and performed plain radiography and chest CT, which revealed elongated bilateral C7 transverse processes, which were thought to be the cause of TOS (Fig. 2). She was diagnosed with true NTOS and the thoracic surgeon did not recommend surgical treatment due to the long period of onset, severe atrophy, and the lack of recent exacerbation of symptoms. A month later, the symptoms of true NTOS did not show any further deterioration.
Congenital anomalies of bony, fibrous, or muscular structures are often reported as etiologies of NTOS and can be associated with traumatic or functional causes. Bony anomalies, including cervical ribs and elongated C7 transverse processes, are reported to account for 30% of NTOS cases [8]. Cervical ribs are known to have a prevalence of 0.5% to 2%, but they are rarely the cause of NTOS. Their prevalence in true NTOS, which requires a thorough documentation of objective findings of nerve compression for diagnosis, is even rarer, with only 1 in 20,000 to 80,000 cervical ribs leading to true NTOS [8]. To our knowledge, the prevalence of elongation of the C7 transverse process has not yet been clearly reported. However, when clinical and electrodiagnostic findings suggest brachial plexopathy at the lower trunk level, the possibility of true NTOS caused by bony abnormalities increases [8]. In our case, the elongation of the bilateral C7 transverse processes was confirmed by radiologic evaluations, in which the right transverse process was slightly longer than the left, corresponding to the symptoms and signs of true NTOS found only on the patient’s right side. Because true NTOS, an extremely rare disease, exhibits clinical symptoms similar to those of CTS, ulnar neuropathy, and cervical radiculopathies, it is often misdiagnosed, as in our case. If a patient exhibits problems only in the hand, as reflected in our case, NTOS is often mistaken for CTS [6]. A physical examination of the median and ulnar nerves and special examinations for NTOS, such as the Roos stress test and Adson’s test, are helpful for establishing a clinical suspicion of NTOS. In the Roos stress test, the patient sits with full external rotation and 90° abduction of the shoulder joint and 90° flexion of the elbow joint, and repeatedly opens and closes the hand for 3 minutes. The provocation of pain or paresthesia is defined as a positive test finding (sensitivity, 52%-84%; specificity, 30%-100%). In Adson’s test, after the patient fully extends the elbow joint on the symptomatic side, the examiner palpates the radial pulse, as the patient turns the neck toward the symptomatic side while holding a deep breath. A change in the radial pulse indicates a positive test (sensitivity, 79%; specificity, 74%-100%) [1,3]. In order to exclude other possibilities from the differential diagnosis and confirm true NTOS, an electrodiagnostic study is essential [3]. In nerve conduction studies, reduced median CMAP, ulnar and MABC SNAP amplitudes, and normal median SNAP and ulnar CMAP amplitudes indicate chronic axonal loss at the lower trunk level of the brachial plexus [6,9]. On needle electromyography, denervation potentials in C8- and T1-innervated muscles (dominant T1), including the APB, ADM, FDI, and other hand intrinsic muscles, are typical findings of true NTOS [6,9]. Abnormal MABC SNAP (sensitivity, 95%) and abnormalities in the APB (sensitivity, 97%) on needle EMG are highly sensitive for true NTOS [7,10]. In our case, the electrodiagnostic findings were compatible with those of true NTOS. In addition, this case is noteworthy due to the mild decrease in the right ulnar CMAP amplitude, abnormalities in the C8-innervated muscles in needle electromyography, and evidence of chronic denervation, such as high MUAP amplitudes instead of abnormal spontaneous activities, reflecting the patient’s long period of onset and severe atrophy. Based on the electrodiagnostic results and clinical findings that revealed no deterioration of symptoms in the past 3 months, it could be inferred that the denervation was not an ongoing process; thus, conservative treatment was recommended.
This is a rare case in which true NTOS was diagnosed 5 years after the onset of symptoms, during comprehensive rehabilitation that the patient received for left hemiplegia due to an unrelated cause. True NTOS is often overlooked or mistaken for CTS, thus delaying the diagnosis and inevitably worsening the outcomes. In addition to detailed history-taking and a physical examination, an electrodiagnostic study is crucial to detect brachial plexopathy and rule out other possibilities in the differential diagnosis, and radiologic evaluations may also be helpful.
Fig. 1.
Atrophic changes in (A) right abductor pollicis brevis, abductor digiti minimi, and (B) right first dorsal interosseous muscles. (C) Positive Wartenberg’s sign in the right hand. We received the patient’s consent form about publishing all photographic materials.
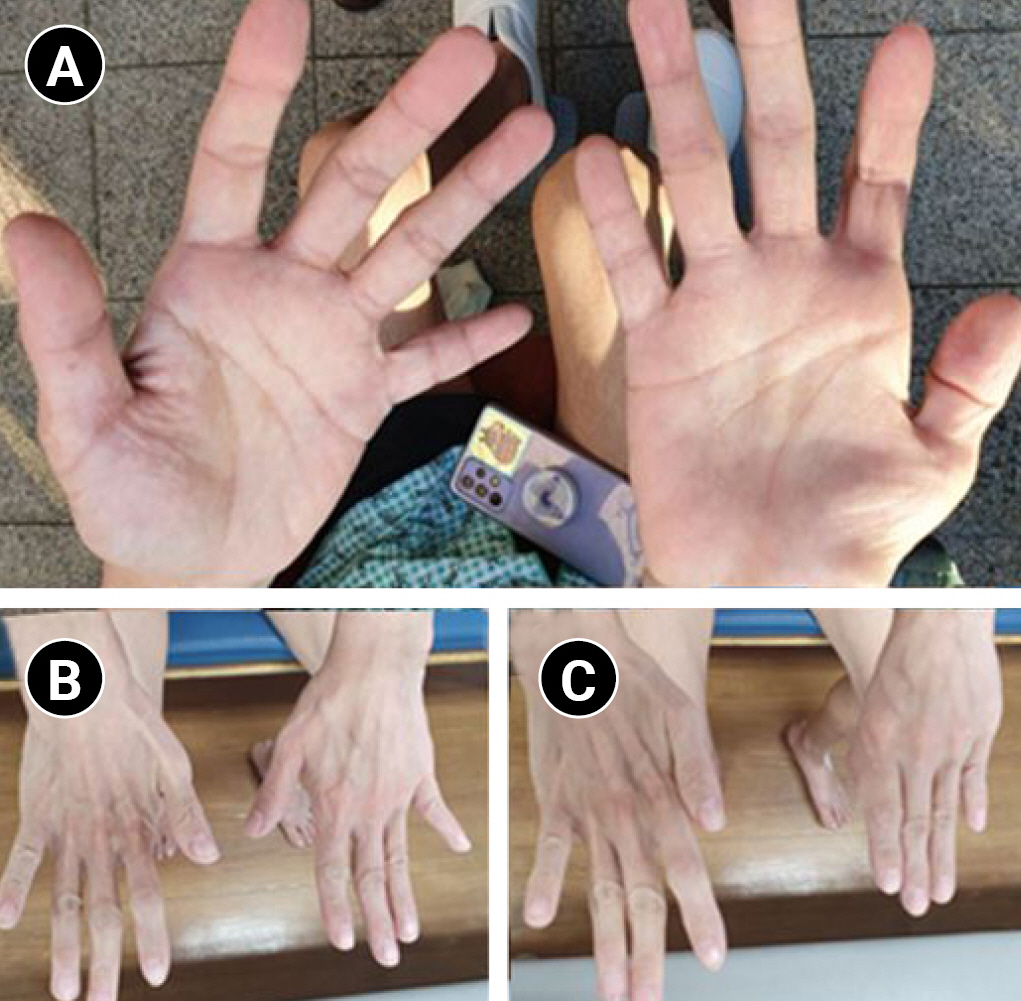
Fig. 2.
Elongation of bilateral C7 transverse processes confirmed by plain radiography (A) and chest computed tomography (B).
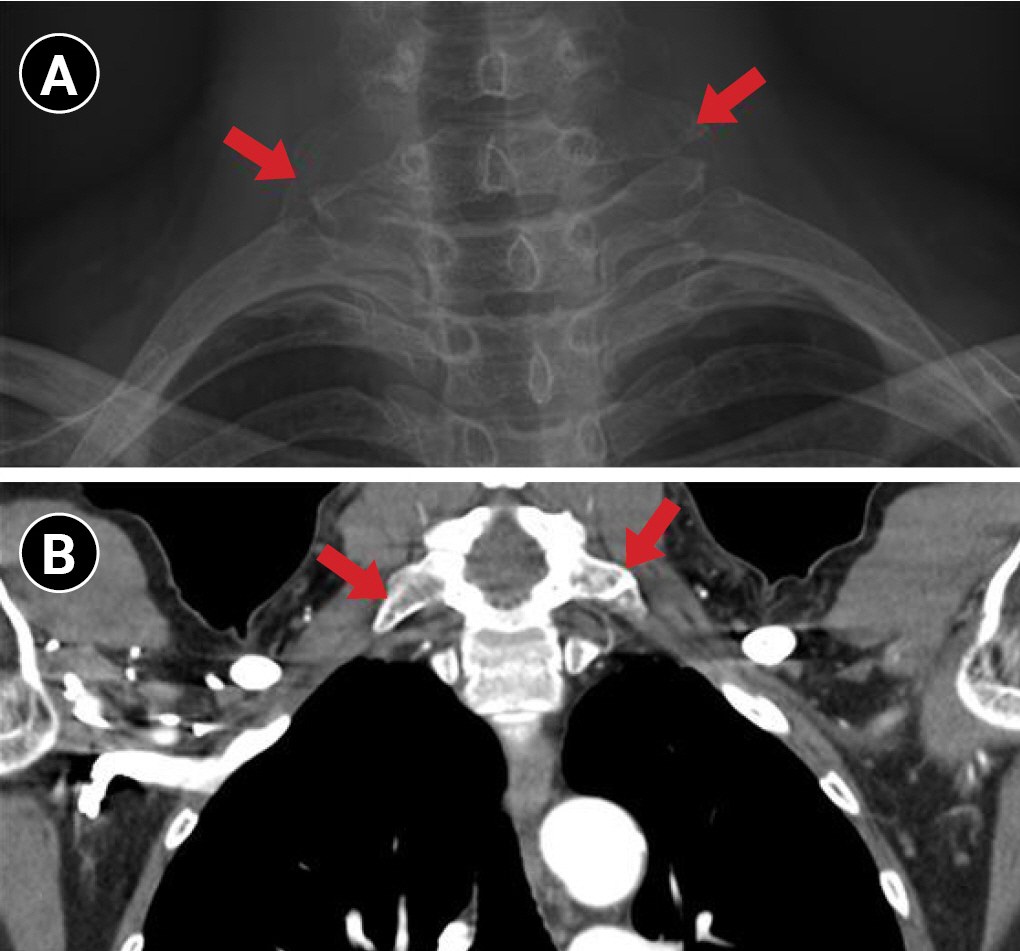
Table 1.
Results of Nerve Conduction Studies
| Nerve | Stimulation site | Recording site | Latency (ms) | Amplitude | Conduction velocity (m/s) |
|---|---|---|---|---|---|
| Motor nerve conduction | |||||
| Rt. median | Wrist | APB | NR* | NR* | NR* |
| Elbow | APB | NR* | NR* | NR* | |
| Lt. median | Wrist | APB | 3.1 | 9.8 | 60 |
| Elbow | APB | 7.1 | 7.5 | ||
| Rt. ulnar | Wrist | ADM | 3.0 | 5.9 | 56 |
| Below elbow | ADM | 7.1 | 5.6 | 77 | |
| Above elbow | ADM | 8.4 | |||
| Lt. ulnar | Wrist | ADM | 2.0 | 8.7 | 68 |
| Below elbow | ADM | 5.4 | 8.4 | 71 | |
| Above elbow | ADM | 6.8 | 7.8 | ||
| Rt. radial | Forearm | EIP | 2.5 | 3.4 | NA |
| Lt. radial | Forearm | EIP | 2.0 | 2.8 | NA |
| Sensory nerve conduction | |||||
| Rt. median | Wrist | Third digit | 3.1 | 25 | NA |
| Lt. median | Wrist | Third digit | 2.8 | 26 | NA |
| Rt. ulnar | Wrist | Fifth digit | 2.9 | 8* | NA |
| Lt. ulnar | Wrist | Fifth digit | 3.0 | 38 | NA |
| Rt. radial | Forearm | Snuffbox | 2.7 | 31 | NA |
| Lt. radial | Forearm | Snuffbox | 1.9 | 25 | NA |
| Rt. MABC | Elbow | Forearm | 2.3 | 12 | NA |
| Lt. MABC | Elbow | Forearm | 2.1 | 18 | NA |
| Rt. LABC | Elbow | Forearm | 2.5 | 32 | NA |
| Lt. LABC | Elbow | Forearm | 1.9 | 32 | NA |
Amplitudes are measured in millivolts (mV, motor) and microvolts (μV, sensory).
Rt., right; APB, abductor pollicis brevis; NR, no response; Lt., left; ADM, abductor digiti minimi; EIP, extensor indicis proprius; NA, not applicable; MABC, medial antebrachial cutaneous; LABC, lateral antebrachial cutaneous.
Table 2.
Results of Needle Electromyography
References
1. Laulan J, Fouquet B, Rodaix C, Jauffret P, Roquelaure Y, Descatha A: Thoracic outlet syndrome: definition, aetiological factors, diagnosis, management and occupational impact. J Occup Rehabil 2011;21:366-373.



2. Povlsen S, Povlsen B: Diagnosing thoracic outlet syndrome: current approaches and future directions. Diagnostics (Basel) 2018;8:21.



3. Hooper TL, Denton J, McGalliard MK, Brismée JM, Sizer PS: Thoracic outlet syndrome: a controversial clinical condition. Part 1: anatomy, and clinical examination/diagnosis. J Man Manip Ther 2010;18:74-83.



4. Illig KA, Rodriguez-Zoppi E: How common is thoracic outlet syndrome? Thorac Surg Clin 2021;31:11-17.


6. Le Forestier N, Moulonguet A, Maisonobe T, Léger JM, Bouche P: True neurogenic thoracic outlet syndrome: electrophysiological diagnosis in six cases. Muscle Nerve 1998;21:1129-1134.


7. Tsao BE, Ferrante MA, Wilbourn AJ, Shields RW: Electrodiagnostic features of true neurogenic thoracic outlet syndrome. Muscle Nerve 2014;49:724-727.


8. Chang MC, Kim DH: Essentials of thoracic outlet syndrome: a narrative review. World J Clin Cases 2021;9:5804-5811.










