Guillain-Barré Syndrome Following the ChAdOx1-S/nCoV-19 Vaccine: A Case Report
Article information
Abstract
Serious neurological complications following coronavirus disease 2019 (COVID-19) vaccination, such as cerebral venous sinus thrombosis or acute disseminated encephalomyelitis, have rarely been reported. Here, we report a case of Guillain-Barré syndrome (GBS) that occurred within 2 weeks of receiving the AstraZeneca ChAdOx1-S/nCoV-19 vaccine. A 61-year-old man presented with weakness and hypesthesia of the legs 9 days after the first dose of the ChAdOx1-S vaccine. The patient progressed to respiratory failure and severe quadriparesis. Nerve conduction studies showed markedly reduced amplitudes of compound muscle action potentials and sensory nerve action potentials in both upper and lower extremities, without definitive evidence of demyelination. The patient was diagnosed with axonal GBS and received intravenous immunoglobulin treatment, with a poor response. This is a rare case of GBS after AstraZeneca ChAdOx1-S/nCoV-19 vaccination in Korea. Physicians should be aware of this rare but serious complication of the COVID-19 vaccine to effectively manage such a situation in clinical practice.
Introduction
The coronavirus disease 2019 (COVID-19) pandemic has been ongoing for more than a year and a half, and COVID-19 vaccines obtained through emergency use approval are becoming an important factor in the stabilization of the pandemic [1]. As of July 26, 2021, approximately 16.9 million of the Korean population had received the COVID-19 vaccine.
In Korea, from February 2021, reports of various side effects related to the vaccine started surfacing, mainly among healthcare workers who were administered the vaccine at the beginning of the immunization program [2,3]. Recently, serious complications such as thromboembolic events arising from the Oxford AstraZeneca ChAdOx1nCov-19 vaccine and myocarditis from the Pfizer-BioNTech COVID-19 vaccine have been reported [3].
As more and more people are being vaccinated in order to achieve herd immunity within the general population, reports of rare but serious adverse events are increasing, and the demand for the analysis of vaccine related disease and causality is increasing. Guillain-Barré syndrome (GBS) is one of the rare adverse events reported after COVID-19 vaccine and has been reported in some parts of the world [4]; however, no such case has been reported in Korea to date. To the best of our knowledge, we report a rare case of GBS manifesting as ascending quadriparesis 2 weeks after receiving the Oxford AstraZeneca COVID-19 vaccine in Korea.
The study was approved by the Ethics Committees of the Inje University Busan Paik Hospital. Informed consent was obtained from the patient.
Case Report
A 61-year-old male patient without significant comorbidities presented with progressive paraparesis. The patient had nausea, vomiting, headache, and dizziness at presentation which started 3 days ago. There was no known history of COVID-19 exposure. The COVID-19 reverse-transcriptase polymerase chain reaction test performed nine months prior to this presentation was negative. He received his first dose of the Oxford AstraZeneca ChAdOx1-S/nCoV-19 vaccine nine days prior to paraparesis presentation. After the initial paralysis of the lower extremities, muscle strength in the arms also began to decrease gradually, and on the day before the visit to the hospital, the patient was having difficulty in speaking and swallowing accompanied by respiratory problems. At the time of his arrival to the emergency room, respiratory failure had progressed, and thus, intubation was performed.
Neurological examination revealed dysarthria and dysphagia. There was severe weakness in the upper extremities (Medical Research Council grade 2/2) and the lower extremities (grade 0-1/0-1). Deep tendon reflexes in the upper and lower extremities were absent.
Thereafter, routine blood investigations, including serological tests, assays for paraneoplastic markers, and cerebrospinal fluid (CSF) analysis, were performed, which ruled out several etiologies. CSF examination revealed albuminocytologic dissociation (white blood cell [WBC] count, 0/mm3; protein, 87.6 mg/dL). Laboratory tests revealed normal WBC counts, C-reactive protein levels, and erythrocyte sedimentation rate, thus pointing toward an absence of inflammation. The screening tests for systemic vasculitis were also normal. No paraproteins were detected using serum protein electrophoresis. Testing for respiratory viruses, such as influenza and COVID-19, that can cause respiratory failure was carried out and yielded negative results. Brain computed tomography scan showed no significant abnormality.
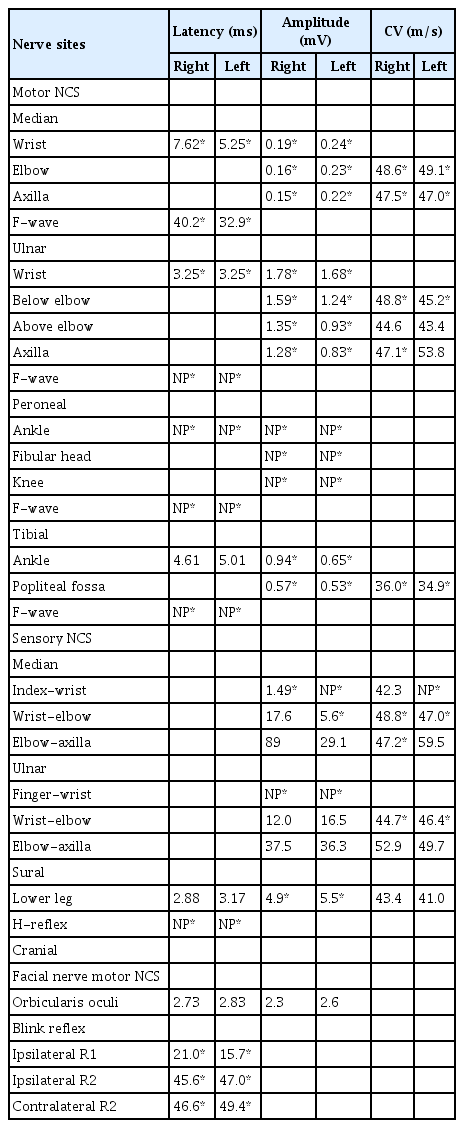
Nerve conduction studies (NCS) performed on the sixth day of initial presentation revealed acute motor sensory axonal neuropathy (Table 1). There was mild slowing of conduction velocities and severely decreased compound muscle action potentials without definite evidence of demyelination in both forearms and legs. Sensory NCS showed decrease in sensory nerve action potential amplitude and conduction velocities of both the upper extremities and sural nerves. There was an absence of the H-reflex in both lower limbs and an absence or delay of F-waves in all 4 extremities, along with a bilateral delayed response in the blink reflex pathway. All the above findings led to the diagnosis of axonal GBS.
Treatment with intravenous immunoglobulins was started, but the symptoms gradually worsened and progressed to quadriplegia. One week after admission, bilateral abducens nerve palsy progressed to bilateral complete ophthalmoplegia, facial diplegia, and bilateral ptosis.
Autonomic nervous system symptoms presented with cardiovascular dysautonomia, including fluctuations in blood pressure and heart rate, and gastrointestinal dysmotility leading to constipation.
Antiganglioside antibody analysis, including immunoglobulin (Ig)G and IgM antibodies against gangliosides GM1, GD1b, and GQ1b revealed negative results.
Two months after the onset of symptoms, severe ptosis, ophthalmoplegia, and bilateral facial palsy resolved moderately, and quadriparesis also began to improve gradually (upper extremities: grade 1-2/1-2; lower extremities: grade 1/1). Tracheostomy with mechanical ventilation was still maintained.
Discussion
GBS has been reported as one of the neurological manifestations that can occur in patients with COVID-19 [5]; however, it has rarely been reported since the initiation of COVID-19 vaccination program worldwide [6]. In Korea, healthcare workers were the first to receive COVID-19 vaccines, and starting in February 2021, local or systemic adverse effects related to the vaccine, such as pain, fever, chills, and myalgia, were reported in some cases [2]. However, reports on rare and serious complications such as thrombosis and cardiomyositis are scarce. As of July 26, 2021, approximately 16.9 million Korean people (32.9%) had been vaccinated with the first dose of the COVID-19 vaccine, and one reason behind the fewer number of reports on side effects may be related to the fact that a major part of the population is not vaccinated. Another reason may be limited interest in serious complications or difficulty in explaining the relationship between the vaccine and the complications.
Axonal GBS, one of the subtypes of GBS, has more severe symptoms and poorer prognosis than acute inflammatory demyelinating polyradiculopathy, and is mainly related to Campylobacter jejuni infection [7]. Neurological adverse effects that may occur after immunization are mainly demyelinating diseases, and association between influenza vaccines and GBS has been reported [7]. Although the reported risk of GBS after influenza vaccine is moderate, the vaccine is generally recommended because it outweighs the risk of H1N1 influenza morbidity and mortality [8]. Some studies suggest that post-vaccination demyelinating events are likely to trigger clinical disease expression in individuals who already have an underlying disease process [9]. Even in cases of GBS, there are conflicting opinions regarding the association with vaccination. In many studies conducted in the early days of the pandemic, it was noted that the antigenicity of severe acute respiratory syndrome coronavirus 2 (SARS-COV-2) itself is not involved in the causation of GBS, and therefore, ganglioside antibody is not found in most GBS cases after COVID-19 infection [5]. In addition, in post-vaccination GBS, ganglioside antibody in post-infectious GBS was not found. In the present case, GM1, GD1b, and GQ1b ganglioside antibodies were not detected.
However, recently, it has been shown that the COVID-19 spike protein binds to the glycolipoprotein-containing sialic acid present in coronavirus as well as peripheral nerve myelin, suggesting the possibility of cross-reactivity between SARS-COV-2 and peripheral nerves [10]. Although it is difficult to explain the mechanism of development of GBS following COVID-19 vaccination [4], if GBS develops within a few days after inoculation and cannot be explained by any other disease, the relationship between GBS and vaccine cannot be completely ruled out.
In this case, it was difficult to determine whether GBS was an isolated occurrence caused by an asymptomatic infection or related to the COVID-19 vaccine. However, in this case, there were no respiratory or gastrointestinal symptoms before onset, and no significant abnormalities were found in laboratory tests carried out for diagnosing the other possible causes. Therefore, we speculate that vaccination may have acted as a trigger in the development of GBS, as there was a history of receiving ChAdOx1-S/nCoV-19 vaccination 2 weeks prior to the onset, and the likelihood of other causes was low.
The annual incidence rate of GBS in Korea was 1.48 patients per 100,000 population through a nationwide epidemiological study from 2010 to 2016 [11]. Also, according to the Korean National Health Insurance Service claims data, a causal relationship between the occurrence of GBS within 6 weeks (42 days) and the vaccine was not found in the elderly who were mainly vaccinated against influenza [12]. Since it is still difficult to explain the causal relationship between GBS and the COVID-19 vaccine, there are not enough studies on the report itself or the relationship between the occurrence of GBS after the COVID-19 vaccine in Korea and around the world. Therefore, since more than 80% of the total population has completed the first dose of vaccination by the end of October in Korea, it is possible to study the causal relationship between GBS and the COVID-19 vaccine in a follow-up study based on nationwide vaccination, and the difference between the influenzae vaccine and the COVID-19 vaccine in the occurrence of GBS can also be compared.
The classification of GBS subtypes or NCS in patients who developed GBS after the COVID-19 vaccine has not yet been established. It can be seen that both the demyelinating type and the axonal type occur in GBS after COVID-19 infection [5], and it can be seen that both the demyelinating type or axonal type occurred in GBS that occurred after the corona 19 vaccination [13]. Future studies on the neurophysiology of GBS associated with the COVID-19 vaccine will be needed through a study that recruited patients with a high causal relationship with the COVID-19 vaccine.
Although vaccines approved for emergency use have some rare but serious complications in addition to the minor side effects, the benefits far outweigh these risks; therefore, vaccination programs are continuing around the world [1].
Because it targets the general population without disease, serious complications such as GBS may occur that require intensive care unit treatment due to neuromuscular respiratory failure. Efforts should be made to keep the immunization program stable through a good medical emergency management system.
In conclusion, although it is not yet possible to explain the direct relationship between the COVID-19 vaccine and GBS, this report raises interest in the occurrence of such diseases as a complication of vaccines. Further studies are needed in this area to thoroughly explore this relationship and determine the possible link between COVID-19 vaccination, GBS, and the related long-term neurological sequelae. A complete investigation of adverse events associated with COVID-19 vaccines will not only help in gathering accurate information about the vaccine but also guide the clinicians toward a better management of such events in practice.
Notes
Conflict of Interest
No potential conflict of interest relevant to this article was reported.
Acknowledgements
This study was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIT) (No. 2020R1G1A1008446).