 |
 |
- Search
| J Electrodiagn Neuromuscul Dis > Volume 22(2); 2020 > Article |
|
Abstract
Superficial temporal artery-middle cerebral artery (STA-MCA) bypass is a surgical treatment for ischemic stroke caused by large artery occlusion. Intraoperative neuropsychological monitoring (IONM) is useful for ensuring cerebral perfusion. Herein, we report 5 cases of patients with ischemic stroke who underwent STA-MCA bypass with IONM. We investigated whether the improved cerebral perfusion was reflected in real time on the evoked potentials (EP) of IONM. To measure changes in cerebral perfusion, all patients underwent pre- and post-operative perfusion-weighted imaging (PWI). Additionally, functional assessments were performed before and after the surgery. We found that the motor evoked potential (MEP) amplitudes showed a consistent increase at the end of surgery. The somatosensory evoked potential (SSEP) latencies decreased consistently, but this change was not large. SSEP amplitudes showed no consistent change. Most of the PWI findings and functional assessments of our patients also improved after the surgery. After bypass surgery, MEP showed more reliable and consistent changes than SSEP.
Revascularization treatment methods for ischemic stroke include intravenous thrombolysis, endovascular recanalization, and mechanical thrombectomy [1]. In patients with cerebral infarction caused by occlusion of large arteries such as the middle cerebral artery (MCA) or the internal carotid artery (ICA), superficial temporal artery to middle cerebral artery (STA-MCA) bypass surgery may be considered as a treatment method. This is considered particularly when endovascular recanalization therapy is ineffective and the compensatory blood circulation corresponding to the reduced cerebral blood perfusion does not occur adequately [2].
Monitoring cerebral blood flow is very important during STA-MCA bypass surgery. Sonographic flowmetry and indocyanine green angiography are two methods currently being used to assess blood flow in real time. These tests employ flow-oriented monitoring, and are used in the operating room because they can provide immediate and visual feedback [3]. Pre- and post-operative perfusion weighted magnetic resonance imaging (PWI) is another method that can be used to check the cerebral perfusion and the improved blood flow quantitatively. PWI images are also helpful in checking the pattern of blood flow changes according to the different brain regions [4].
Intraoperative neurophysiological monitoring (IONM) has been widely applied as an important diagnostic tool during cerebrovascular surgeries. With IONM, neurologic integrity and functional status as well as overall perfusion status can be monitored during the entire operation. Patient safety can be ensured by real-time detection of abnormal signs due to insufficient cerebral perfusion or a decrease in local blood. Numerous studies have demonstrated that the application IONM during cerebrovascular surgeries could prevent neurological damage [5]. However, only a few reports of the application of IONM during STA-MCA bypass surgery currently exist. Moreover, no report has been presented as to whether the improvement in blood flow seen during direct bypass surgery could be reflected as a change in the evoked potential (EP) on IONM.
This is a case series report of 5 patients who underwent the STA-MCA bypass surgery with IONM. This study protocol was reviewed and approved by the Institutional Review Board (approval No. PSSH0475-201911-HR-004-01).
For each patient, we observed the changes in EP on IONM that occurred between baseline and the end of the operation. The amplitude of the motor evoked potential (MEP) and the latency and amplitude of the somatosensory evoked potential (SSEP) were measured on the lesion side. MEP recorded from the abductor pollicis brevis muscle (APB-MEP) and tibialis anterior muscle (TA-MEP) were measured. For SSEP, median SSEP recorded from the median nerve at the wrist and tibial SSEP recorded from the posterior tibial nerve at the ankle level were measured. Total intravenous anesthesia regimen (propofol, 3-5 ┬Ąg/mL and remifentanil, 3-5 ng/mL) was applied during the surgery. A single bolus of neuromuscular blocking (NMB) agent (rocuronium, 0.4-0.5 mg/kg) was used before intubation. The demographic factors of patients are summarized in Table 1.
We observed the PWI findings before and 1 week after surgery. The region of interest was positioned at the centrum semiovale (CS) and the subcortical level of primary motor and sensory cortex area (SPMS). For functional assessment, we measured the modified Rankin scale (mRS) and the modified Barthel index (MBI) at the same time as the PWI. The results of EP, PWI, and functional assessments are summarized in Table 2.
An 80-year-old female patient who had left MCA occlusion underwent STA-MCA bypass surgery 2 months after stroke onset. She had hypertension, which was a vascular risk factor. Median SSEP showed a decrease in N20 latency and an increase in amplitude, but the degree of change was 2.37% and 7.16%, respectively. For tibial SSEP, P37 latency decreased by 2.3%; however, at the end of the surgery, the amplitude decreased by 31.03%. The amplitude of APB-MEP increased by 1.12%, which was a minute degree of change. The amplitude of TA-MEP increased by 32.56%. PWI revealed that the mean transit time (MTT) at the SPMS and CS had shortened. Functional assessments showed no changes.
A 57-year-old female patient who had right MCA occlusion underwent STA-MCA bypass surgery 2 weeks after stroke onset. She had hypertension and diabetes. Both the median and tibial SSEP showed decreases in latency, but the degree of change was 1.68% and 0.68%, respectively. SSEP amplitudes decreased at the end of the surgery. An increase in amplitude was observed in all the measured MEPs. The amplitude of APB-MEP increased by 79.65% indicating a large amount of change. Meanwhile, the amplitude of TA-MEP increased by only 4.44%, which was a slight increase. In the PWI findings, both SPMS and CS showed the most reduced MTT at 1 week after surgery. Even before the surgery, the patient had an MBI score of 96 and had not experienced a significant decline in daily activities. Thus, the patient was able to resume normal function following the surgery.
A 45-year-old male patient who had left MCA occlusion underwent STA-MCA bypass surgery 2 weeks from the stroke onset. He had diabetes. Both SSEP and MEP were not evoked at the baseline. At the end of the surgery, a small waveform of tibial SSEP was evoked; however, the delay in P37 latency was still severe and the amplitude was small. Median SSEP and all MEPs were not evoked at the end of surgery. PWI conducted at 1 week after surgery revealed improved MTT at the CS (Fig. 1). However, at the SPMS, PWI showed no clear improvement in perfusion. The patient scored a 5 and a 4 on the mRS before 1 week after surgery, respectively. His MBI before surgery was 5. Thereafter, the patient showed an improvement in the functional status by scoring an MBI of 37 at 1 week after surgery.
A 42-year-old male patient who had left ICA occlusion underwent STA-MCA bypass surgery 6 months from the stroke onset. He had hypertension and was a smoker. Median SSEP showed a decrease in N20 latency by 1.85% and an increase in amplitude by 11.94%. For tibial SSEP, P37 latency decreased by 6.60%. In contrast, the amplitude decreased, but the degree of change was small. The amplitude of APB-MEP increased by 362.92%, and TA-MEP increased by 10.46%. On postoperative PWI, MTT at the CS and the SPMS showed an obvious reduction. Functional assessments showed that his mRS score and MBI before surgery were 3 and 70, respectively, and there were no changes in these scores 1 week after surgery.
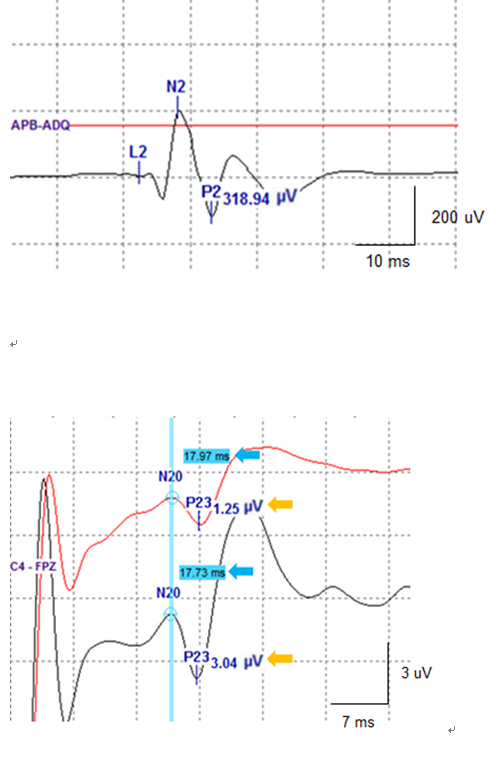
A 52-year-old female patient who had right MCA occlusion underwent STA-MCA bypass surgery 6 months from the stroke onset. She had hypertension, diabetes, and coronary artery disease. Both median SSEP and tibial SSEP showed decreases in their latencies and increases in amplitude. Median SSEP showed a considerable change with an increase in amplitude by 143.20%. APB-MEP was not evoked at the baseline. At the end of the surgery, however, we observed a waveform representing a significant change (Fig. 2). TA-MEP also showed a significant change with an increase of 146.13% in amplitude at the end of surgery. The patient scored 3 on the mRS and 70 on the MBI before surgery. At 1 week after surgery, the patientŌĆÖs functional status improved (mRS score, 2 and MBI, 88). Although findings from both EP and functional assessments indicated improvements, PWI showed an increase in MTTs.
With IONM, functional integrity of motor and sensory pathways can be detected in real time during surgery. IONM is distinguished from other real-time blood flow monitoring methods or PWI as it has the advantage of reflecting the patientŌĆÖs neurological and functional states [3]. In this study, we investigated whether the improved cerebral perfusion after vascular anastomosis during STA-MCA bypass surgery was reflected in real time on the EP on IONM.
MEP and SSEP are key IONM modalities that are mainly used for cerebrovascular surgeries. MEP is sensitive as it reflects the functional state of motor pathways and rapidly reacts to neurological deterioration; however, it has the disadvantage of having a high variability [6]. Contrastingly, SSEP reflects the functional state of sensory pathways and is also closely related to other factors such as blood pressure and body temperature as well as overall cerebral blood flow. SSEP generally has a higher specificity and relatively lower sensitivity than MEP. SSEP response to intraoperative events has also been shown to be slower than that of MEP [7]. Due to these characteristics, in case of subcortical ischemia, MEP has a rapid response, but SSEP can display false negatives [8]. On the other hand, according to the anatomical features of the brain motor pathway, lower extremity SSEP may react more sensitively to ischemia than MEP in the case of anterior cerebral artery or an anterior choroidal artery procedures [9]. In addition, since MCA supplies both primary motor and sensory cortices, changes in SSEP can indirectly reflect motor deficit [5]. In most cases, MEP and SSEP are monitored simultaneously. Such multimodal EP monitoring gives the advantage of providing complementary information to physiatrists and surgeons.
IONMŌĆÖs role as a preventive measure for ensuring patient safety is being continuously emphasized. Based on previous reports, our warning criteria for IONM during cerebrovascular surgery are as follows: 1) MEP amplitude is reduced by 50% or more, 2) SSEP amplitude is reduced by 50% or more, or 3) SSEP latency is increased by 10% or more [7,9]. Such warning signals indicate not only neural insults such as blood flow reduction or nerve damage during surgery but also represent significant changes concerning systemic conditions such as hypoxia, hypothermia, hypotension, and anesthesia [5]. On the other hand, little has been known about the change in EP when blood flow improved in patients with ischemia. One previous study reported that the degree of recanalization after mechanical thrombectomy was related to changes of EP and functional outcomes in patients with hyperacute stage stroke [10]. However, there has been no study demonstrating the relationship between improvement of cerebral blood flow and corresponding EP changes after the direct bypass surgery in the subacute stage of stroke patients with large artery occlusion.
In this study, the MEP amplitude showed an average of 50% or more improvement in amplitude. In case 5, APB-MEP was not evoked at baseline. However, at the end of the surgery, APB-MEP was evoked in addition to improvements in other EP findings. In case 3, no MEP and SSEP were evoked at baseline. This patient was transferred from other hospital with left MCA infarction. Upon initial assessment, according to Medical Research Council (MRC), muscle strength in his right upper and lower extremities were 1/5 (Trace). Preoperative EP study was performed 24 hours prior to the surgery, and neither MEP nor SSEP was evoked at the right upper and lower extremities. However, MEP and SSEP were normally evoked in the left upper and lower limbs. Therefore, unobtainable EP on the right side at the baseline study were more likely to be the result of cerebral infarction rather than the possibility of polyneuropathy, such as diabetes. Most MEP and SSEP were not evoked after the surgery, however, at the end of the surgery, although it was a minute change, a waveform was evoked on tibial SSEP.
In our opinion, this SSEP change was associated with the improvement of MTT in CS on PWI performed one week after the surgery. However, there was no sufficient recovery of cerebral blood flow to induce dramatic improvement of EP during surgery.
A comprehensive analysis of the aforementioned EP findings revealed that MEP amplitude shows an immediate response to the improvement in cerebral perfusion after STA-MCA anastomosis. We view that it is because MEP itself has high sensitivity and MEP showed a more dramatic change because the motor tract is mainly supplied by the MCA. All patientsŌĆÖ baseline EP data were gained just before the opening of dura mater. EP data obtained after the skin closure were used as the end of operation data. In this study, baseline EP data were obtained just before the opening of dura mater. This means that the baseline MEP data used in this study had a time interval to minimize the effect of the NMB agent, and thus, the effect of the NMB agent can be excluded in the interpretation of MEP findings. The time taken to obtain baseline EP data from the first train-of-four response was 85.4 minutes (75-101).
On the other hand, SSEP did not show consistent improvements as expected. Consequently, our reasoning is that the intra-operative hemodynamic changes after the vascular anastomosis are not enough to reduce the SSEP latency or increase SSEP amplitude in terms of temporal aspect.
In our study, most MTTs were reduced at 1 week after surgery. In case 5, postoperative PWI showed that MTT was delayed after surgery. PWI findings were in contrast to EP findings and functional assessments, both of which showed improvements. However, judging by the fact that both mRS and MBI results improved, we infer that MTT delay is not necessarily related to a functional change. In a previous study comparing PWI results after STA-MCA bypass surgery, MTT showed gradual improvements in the 1-week and 3-month postoperative PWI studies, and these results were consistent with those of our study [4].
In STA-MCA bypass surgery, multimodal EP monitoring can be used to predict the improvement in brain perfusion intraoperatively. In particular, in this case report, MEPs showed more reliable and consistent changes than did SSEPs during bypass surgery.
Fig.┬Ā1.
Perfusion weighted magnetic resonance imaging (PWI) of case 3. Preoperative PWI showed prolonged mean transit time (MTT) (A). As can be seen, PWI at 1 week after surgery showed that MTT decreased following STA-MCA bypass (B). Region of interest was positioned at the centrum semiovale level.
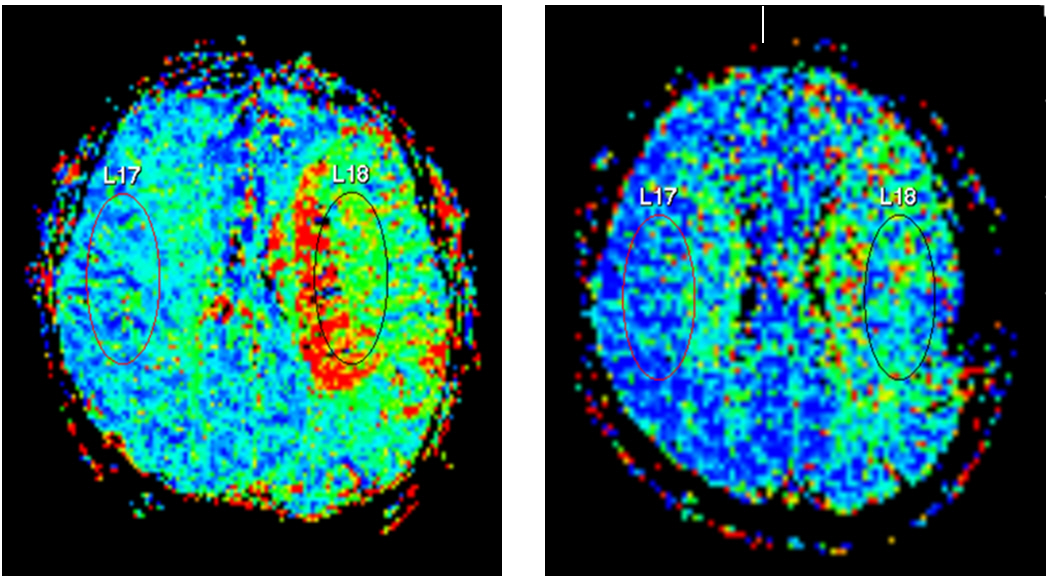
Fig.┬Ā2.
Results of evoked potentials in case 5. Baseline MEP (red line) recorded from the abductor pollicis brevis muscle was not evoked but appeared at the end of surgery (black line) (A). Median SSEP showed a 143% increase in amplitude compared with the baseline (orange arrows), but there was no significant change in N20 latency (blue arrows) (B).
Table┬Ā1.
Characteristics of Patients
Table┬Ā2.
Findings of IONM, PWI, and Functional Assessments
|
Somatosensory evoked potential* |
Motor evoked potential* |
PWIŌĆĀ |
Clinical dataŌĆĀ |
||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
|
Median nerve |
Tibial nerve |
APB | TA | SPMS-MTT (s) | CS-MTT (s) | mRS | MBI | ||||
| N20 (ms) | Amplitude (uV) | P37 (ms) | Amplitude (uV) | Amplitude (uV) | Amplitude (uV) | ||||||
| Pre | 20.65 | 11.73 | 43.5 | 0.87 | 2670 | 1290 | 13.9 | 12.4 | 2 | 88 | |
| Case 1 | Post | 20.16 | 12.57 | 42.5 | 0.6 | 2700 | 1710 | 11.9 | 10.3 | 2 | 88 |
| Δ | -0.0237 | 0.0716 | -0.023 | -0.3103 | 0.0112 | 0.3256 | |||||
| Pre | 20.88 | 0.76 | 37 | 1.53 | 617.86 | 764.69 | 11 | 11.7 | 1 | 96 | |
| Case 2 | Post | 20.53 | 0.74 | 36.75 | 1.1 | 1110 | 798.66 | 10.1 | 9.9 | 0 | 100 |
| Δ | -0.0168 | -0.0263 | -0.0068 | -0.281 | 0.7965 | 0.0444 | |||||
| Pre | NR | NR | NR | NR | NR | NR | 10.1 | 10.7 | 5 | 5 | |
| Case 3 | Post | NR | NR | 66.5 | 0.19 | NR | NR | 10.5 | 9.1 | 4 | 37 |
| Δ | Appeared | Appeared | |||||||||
| Pre | 18.9 | 0.67 | 37.75 | 1.31 | 559.49 | 1530 | 13.5 | 12 | 3 | 70 | |
| Case 4 | Post | 18.55 | 0.75 | 35.26 | 1.26 | 2590 | 1690 | 8.1 | 8.6 | 3 | 70 |
| Δ | -0.0185 | 0.1194 | -0.066 | -0.0382 | 3.6292 | 0.1046 | |||||
| Pre | 17.97 | 1.25 | 36.25 | 1.27 | NR | 674.45 | 6.9 | 6.9 | 3 | 70 | |
| Case 5 | Post | 17.73 | 3.04 | 33.5 | 1.34 | 318.94 | 1660 | 8.2 | 7.1 | 2 | 88 |
| Δ | -0.0134 | 1.432 | -0.0759 | 0.0551 | Appeared | 1.4613 | |||||
IONM, intraoperative neurophysiological monitoring; PWI, perfusion weighted imaging; APB, abductor pollicis brevis; TA, tibialis anterior; SPMS, subcortical level of primary motor and sensory cortex area; CS, centrum semiovale; MTT, mean transit time; mRS, modified Rankin scale; MBI, modified Barthel index; Δ, delta; NR, no response.
References
1. Ko SB, Park HK, Kim BM, Heo JH, Rha JH, Kwon SU, et al: 2019 Update of the Korean Clinical Practice Guidelines of Stroke for Endovascular Recanalization Therapy in Patients with Acute Ischemic Stroke. Neurointervention 2019;14:71-81.



2. Soufiany I, Hijrat KA, Soufiany S, Chen L: Bypass surgery for ischemic stroke caused by intracranial artery stenosis or occlusion. Brain Science Advances 2018;4:49-60.

3. Dengler J, Cabraja M, Faust K, Picht T, Kombos T, Vajkoczy P: Intraoperative neurophysiological monitoring of extracranial-intracranial bypass procedures. J Neurosurg 2013;119:207-214.


4. Li Z, Zhou P, Xiong Z, Ma Z, Wang S, Bian H, et al: Perfusion-weighted magnetic resonance imaging used in assessing hemodynamics following superficial temporal artery-middle cerebral artery bypass in patients with Moyamoya disease. Cerebrovasc Dis 2013;35:455-460.


5. Deletis V, Shils JL: Neurophysiology in neurosurgery : a modern intraoperative approach, Amsterdam ; Boston: Academic Press, 2002.
6. Neuloh G, Pechstein U, Cedzich C, Schramm J: Motor evoked potential monitoring with supratentorial surgery. Neurosurgery 2004;54:1061-1070; discussion 1070.


7. Staarmann B, O'Neal K, Magner M, Zuccarello M: Sensitivity and Specificity of Intraoperative Neuromonitoring for Identifying Safety and Duration of Temporary Aneurysm Clipping Based on Vascular Territory, a Multimodal Strategy. World Neurosurg 2017;100:522-530.


8. Holland NR: Subcortical strokes from intracranial aneurysm surgery: implications for intraoperative neuromonitoring. J Clin Neurophysiol 1998;15:439-446.


-
METRICS

-
- 1 Crossref
- Scopus
- 2,967 View
- 51 Download
- Related articles in J Korean Assoc EMG Electrodiagn Med






